 The value of 2 arises from the Dirac equation, a fundamental equation connecting the electron's spin with its electromagnetic properties, and the correction of 0.002319., called the anomalous magnetic dipole moment of the electron, arises from the electron's interaction with virtual photons in quantum electrodynamics. One of the triumphs of the theory of quantum electrodynamics is its accurate prediction of the electron g-factor, which has been experimentally determined to have the value 2.002319. The g-factor is an essential value related to the magnetic moment of the subatomic particles and corrects for the precession of the angular momentum. 
Where the dimensionless quantity g is called the g-factor. The intrinsic magnetic moment μ of a particle with charge q, mass m, and spin s, is The resulting geometric factor is called the Lande g-factor. The persistent early spectroscopists, such as Alfred Lande, worked out a way to calculate the effect of the various directions of angular momenta. This factor is called the electronic spin g-factor. The factor of two multiplying the electron spin angular momentum comes from the fact that it is twice as effective in producing magnetic moment. The magnetic energy of an electron is approximately twice what it should be in classical mechanics. 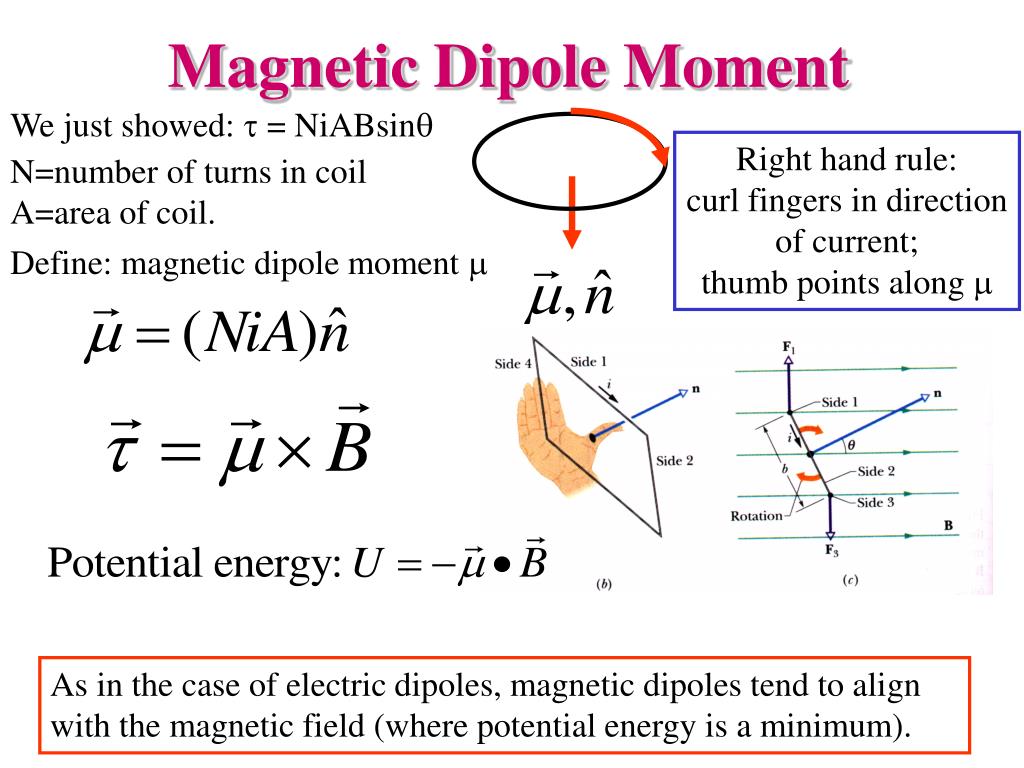
The electron exists in a magnetic field which exerts a torque opposing its alignment creating a potential energy that depends on its orientation with respect to the field. For magnetic dipoles, the dipole moment points from the magnetic south to the magnetic north pole. A rotating electrically charged body in classical electrodynamics causes a magnetic dipole effect creating magnetic poles of equal magnitude but opposite polarity like a bar magnet. The electron is a negatively charged particle with angular momentum. Safe Weighing Range Ensures Accurate Results Explanation of magnetic moment 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
